Organic compounds that contain the –OH group. In systematic chemical nomenclature alcohol names end in the suffix -ol. Examples are methanol, CH3OH, and ethanol, C2H5OH. Primary alcohols have two hydrogen atoms on the carbon joined to the –OH group (i.e. they contain the group –CH2–OH); secondary alcohols have one hydrogen on this carbon (the other two bonds being to carbon atoms, as in (CH3)2CHOH); tertiary alcohols have no hydrogen on this carbon (as in (CH3)3COH): see formulae. The different types of alcohols may differ in the way they react chemically. For example, with potassium dichromate(VI) in sulphuric acid the following reactions occur:
primary alcohol → aldehyde → carboxylic acid
secondary alcohol → ketone
tertiary alcohol – no reaction
Other characteristics of alcohols are reaction with acids to give esters and dehydration to give alkenes or ethers. Alcohols that have two –OH groups in their molecules are diols (or dihydric alcohols), those with three are triols (or trihydric alcohols), etc.
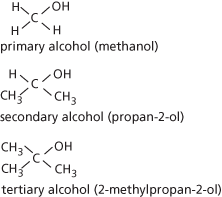
Alcohols
https://www.acdlabs.com/iupac/nomenclature/79/r79_202.htm Information about IUPAC nomenclature
- zero element
- zero energy state
- zero error
- zero function
- zero g(ravity)
- zero growth
- zero IF
- zero interest rate
- zero-length spring
- zero level
- zero matrix
- zero(of a function)
- zero order
- zero order reaction
- zero padding
- zero-point energy
- zero population growth
- zero potential
- zero-rated
- zeros(and poles)
- zero-sum game
- zero suppression
- zeroth degree formula
- zeroth law of thermodynamics
- zero-trip loop